Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
 Ni migration through crushed rock media
Ni migration through crushed rock mediaTanaka, Tadao; Sakamoto, Yoshiaki; Mukai, Masayuki; Maeda, Toshikatsu; Nakayama, Shinichi
Radiochimica Acta, 92(9-11), p.725 - 729, 2004/12
Times Cited Count:1 Percentile:10.03(Chemistry, Inorganic & Nuclear)Migration experiments of  Ni for crushed rocks, granite and tuff, were performed under the coexistent condition with a humic acid and a fulvic acid of 0-30 mg/l in concentration, which are Nordic humic substances supplied from International Humic Substance Society. Migration experiments of Ni had been performed by a column system, to investigate migration behavior of Ni through a column packed crushed rock. The Ni concentration in the effluent passed through the column was corresponding to the fractional percentage of Ni complexing with humic substance in influent solution. This result suggests that the Ni complexing with humic substance in influent solution was flowed out from the column without any effective interactions with the rock media. The migration behavior of Ni could be expressed by a migration model taking account of the complexation kinetics of Ni with humic substance in the aqueous phase.
Ni for crushed rocks, granite and tuff, were performed under the coexistent condition with a humic acid and a fulvic acid of 0-30 mg/l in concentration, which are Nordic humic substances supplied from International Humic Substance Society. Migration experiments of Ni had been performed by a column system, to investigate migration behavior of Ni through a column packed crushed rock. The Ni concentration in the effluent passed through the column was corresponding to the fractional percentage of Ni complexing with humic substance in influent solution. This result suggests that the Ni complexing with humic substance in influent solution was flowed out from the column without any effective interactions with the rock media. The migration behavior of Ni could be expressed by a migration model taking account of the complexation kinetics of Ni with humic substance in the aqueous phase.
Tanaka, Tadao; Sakamoto, Yoshiaki; Sawada, Hiroshi; Ogawa, Hiromichi
JAERI-Conf 2003-010, p.134 - 141, 2003/09
We have performed migration experiments of Np(V) and Am(III) for crushed granite, under the coexistent condition with humic acid substance. As for Np, the periodical concentration changes in the breakthrough curve and the migration velocity of Np passed through the column were not affected by the coexistence of the humic substance. As for Am, on the other hand, the periodical concentration changes in the breakthrough curve were affected by the humic substance concentration. The migration behavior of Am passed through the present column system could be expressed by a migration model taking account of the non-equilibrium state.
 O/alcohol mixed media by time-resolved laser-induced fluorescence spectroscopy
O/alcohol mixed media by time-resolved laser-induced fluorescence spectroscopyArisaka, Makoto*; Kimura, Takaumi; Suganuma, Hideo*; Yoshida, Zenko
Journal of Radioanalytical and Nuclear Chemistry, 255(2), p.385 - 389, 2003/02
Times Cited Count:5 Percentile:36.81(Chemistry, Analytical)no abstracts in English
Tanaka, Tadao; Sakamoto, Yoshiaki; Ogawa, Hiromichi
Genshiryoku Bakkuendo Kenkyu, 9(1), p.29 - 34, 2002/09
To obtain structural information on functional groups of humic substances complexing with metal ions, infrared (IR) spectral analysis of the humic substances dissolving in aqueous solutions has been studied by ATR (Attenuated Total Reflection) method. By using the ATR method, IR spectral shifts of humic substance due to complexation with metal ions were observed. The observation provided with information on complexation kinetics, and on characteristics of functional groups depending on solvent properties, such as pH and solute concentrations. These information was difficult to be obtained by conventional FTIR spectroscopy using powdered humic substances. The obtained results show that IR spectral shifts on the functional groups of the humic substances complexed with metal ions could be possibly characterized  by using the ATR-FTIR spectroscopy.
by using the ATR-FTIR spectroscopy.
Matsunaga, Takeshi; Nagao, Seiya*; Takeda, Seiji; Ueno, Takashi; Amano, Hikaru
Dai-43-Kai Kankyo Hoshano Chosa Kenkyu Seika Rombun Shorokushu (Heisei-12-Nendo), p.49 - 50, 2002/03
no abstracts in English
Takahashi, Yoshio*; Kimura, Takaumi; Minai, Yoshitaka*
Geochimica et Cosmochimica Acta, 66(1), p.1 - 12, 2002/01
Times Cited Count:63 Percentile:73.51(Geochemistry & Geophysics)no abstracts in English
Nagao, Seiya; Nakaguchi, Yuzuru*; Suzuki, Yasuhiro*; Hiraki, Keizo*; Fujitake, Nobuhide*; Ogawa, Hiromichi
Proceedings of International Conference on the Biogeochemistry of Trace Elements (ICOBTE2001), P. 666, 2001/00
no abstracts in English
Takahashi, Yoshio*; Kimura, Takaumi; Kato, Yoshiharu; Minai, Yoshitaka*
Environmental Science & Technology, 33(22), p.4016 - 4021, 1999/00
Times Cited Count:46 Percentile:74.45(Engineering, Environmental)no abstracts in English
G.Meinrath*; Kato, Yoshiharu; Kimura, Takaumi; Yoshida, Zenko
Radiochimica Acta, 84(1), p.21 - 29, 1999/00
no abstracts in English
Kato, Yoshiharu; Kimura, Takaumi; Yoshida, Zenko; Shirasu, Noriko
Radiochimica Acta, 82, p.63 - 68, 1998/00
no abstracts in English
Tanaka, Tadao; Nagao, Seiya; Sakamoto, Yoshiaki; Onuki, Toshihiko; S.Ni*; *
Journal of Nuclear Science and Technology, 34(8), p.829 - 834, 1997/08
Times Cited Count:15 Percentile:74.12(Nuclear Science & Technology)no abstracts in English
*; Sato, Seichi*; Ohashi, Hiroshi*; Sakamoto, Yoshiaki; Onuki, Toshihiko; Nagao, Seiya; Muraoka, Susumu
Journal of Nuclear Materials, 248, p.328 - 332, 1997/00
Times Cited Count:37 Percentile:90.98(Materials Science, Multidisciplinary)no abstracts in English
Sakamoto, Yoshiaki; Nagao, Seiya; Tanaka, Tadao
JAERI-Research 96-055, 14 Pages, 1996/10
no abstracts in English
 Co and
Co and  Am
AmTanaka, Tadao;
Radioisotopes, 44(2), p.99 - 102, 1995/02
no abstracts in English
Sakamoto, Yoshiaki; Nagao, Seiya; Onuki, Toshihiko; ; *; Sato, Seichi*; Ohashi, Hiroshi*
Materials Research Society Symposium Proceedings, Vol.353, 0, p.997 - 1004, 1995/00
no abstracts in English
 Co,
Co,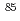 Sr,
Sr, Cs,
Cs, Np and
Np and  Am on soil under coexistence of humic acid; Effects of molecular size of humic acid
Am on soil under coexistence of humic acid; Effects of molecular size of humic acidTanaka, Tadao;
Materials Research Society Symposium Proceedings, Vol.353, 0, p.1013 - 1020, 1995/00
no abstracts in English
 Co,
Co, 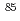 Sr and
Sr and  Cs in coastal sandy soil
Cs in coastal sandy soilTanaka, Tadao; N.Shiwei*
JAERI-M 93-185, 17 Pages, 1993/10
no abstracts in English
Yoshida, Zenko; Aoyagi, Hisao; *
Z. Anal. Chem., 340, p.403 - 409, 1991/00
no abstracts in English
*; *; *; ;
J.Macromol.Sci.,Part A, 23(9), p.1125 - 1135, 1986/09
no abstracts in English